
ASX cannabis stocks are gaining momentum, stimulated by significant developments in the global regulatory environments in recent months.
On the cusp of a potential rerate is $29M capped Neurotech International Limited (ASX: NTI), a stock with imminent newsflow.
NTI is a medical cannabis company focused on the clinical applications of unique cannabis strains to treat neurological disorders.
The last month has been an extremely rewarding period for the company, with stage 1 clinical product formulation and development studies commencing in mid-November, and an outlook to enter into clinical trials in the March quarter of 2021.
Management has also been able to report that further preliminary results of in-vitro human brain cell studies using its proprietary DOLCE/NTI leads, indicated that they can regulate multiple neuronal pathways that are directly involved in cell repair and rejuvenation.
This followed successful preliminary results of in-vitro studies in human neuronal cell studies, positioning the clinical product development program to optimise and develop the most suitable product for phase 1 clinical trial program.

The significance of the group’s achievements hasn’t been missed by investors with the company’s shares surging 130% to a high of 6.6 cents in the last fortnight, a level it hasn’t traded at for more than two years.
Global acceptance of cannabis growing
Neurotech’s recent success in this high profile industry has been driven by both individual and broader factors, as well as the company’s capacity to service high volume markets.
Clinical studies are assessing the neuro-protective, anti-inflammatory and neuro-modulatory activities of its proprietary DOLCE/NTI cannabis strains which include CBDA, CBDP and CBDB.
The strains are unique in that they contain minimal THC (<0.3%), which may result in a less onerous regulatory pathway to commercialisation (any cannabis strain with a THC level above 0.3% is regulated as a narcotic).
These developments are occurring during a time of increasing medical recognition of the benefits of medical cannabis in treating neurological conditions and assisting with mental health problems.
Regulatory authorities are also undergoing increased acceptance of the benefits of CBD, such that previous restrictions that made the drug difficult to access have been lifted and/or eased in many global jurisdictions, including the US, Canada, the European Union and indeed Australia.
This has prompted analysts to revisit forecasts regarding the industry’s growth profile in coming years, with such significant global developments rousing these numbers to increase further.
A significant drain on government health systems, both in human resources and the cost of development and supply of devices for neurological disorders suggests that regulatory bodies will increasingly embrace new technologies that are safe, efficient and less taxing on public medical services.
Neurotech to also benefit from home-based device boom
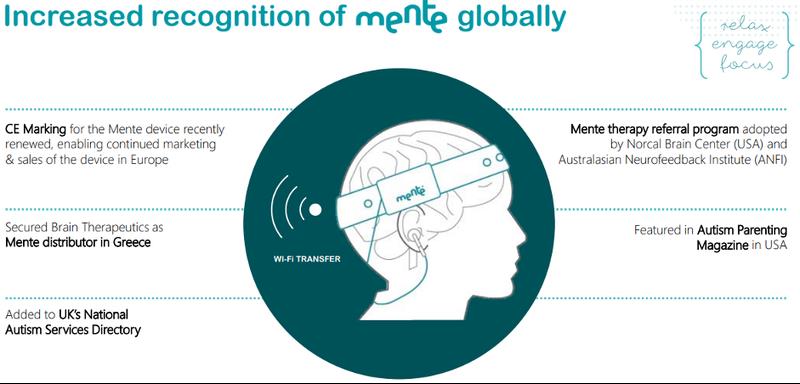
In addition, Neurotech is also commercialising Mente, the world’s first home therapy that is clinically proven to increase engagement and improve relaxation in children on the autism spectrum with elevated Delta band brain activity.
This therapeutic technology in home element is particularly interesting in light of the success of numerous ASX-listed and global enterprises that have met the demand for home use services and devices due to mobility restraints resulting from COVID.
Mente uses personalised audio feedback and earphones to gently guide the brain into a more relaxed state.
It is a soft, easy to use and portable headband that was developed to help those on the autism spectrum better self-regulate attention and mood, which has been based on autism research and a successful clinical trial.
Mente’s daily 40-minute sessions are personalised to the user’s current brain activity and have been designed to minimise any disruption to their daily routine, allowing them to continue with quiet activities (reading, drawing, school work etc) whilst still receiving therapeutic benefits.
This makes Mente perfect for use both at home and within an educational setting.
DOLCE/NTI leads more effective than use of CBD alone
Further data has demonstrated the potential uniqueness of the DOLCE/NTI leads in their mode of action and their differentiation when compared to CBD alone.
Neurotech has been collaborating with high profile medical research organisations in completing in-vitro human brain cell studies using its proprietary DOLCE/NTI cannabis leads, with subsequent results proving very promising.
The studies, conducted at Monash University, University of Wollongong and RMIT University are assessing the neuro-protective, anti-inflammatory and neuro-modulatory activities of the proprietary DOLCE/NTI cannabis leads which include, CBDA, CBDP and CBDB.
Studies have indicated that the DOLCE/NTI leads regulate multiple neuronal pathways which are directly involved in cell repair and rejuvenation.
These leads indicate significant increased potency in repairing neuronal cells when compared to CBD alone.
More importantly, concentrations five times lower of the DOLCE/NTI leads are needed to achieve these results when compared to CBD alone (2ug/ml versus 10ug/ml respectively).
DOLCE/NTI leads work through a novel (different to CBD) mechanism, and they were shown to work through the powerful anti-inflammatory enzyme known as Arginase-1.
Conversely, CBD alone did not produce any significant anti-inflammatory properties.
DOLCE/NTI leads also increased the presence of beta-tubulin, an essential protein in the maintenance and healthy survival of brain cells, while CBD did not produce an increase.
Beta-tubulin is a vital protein in the management of a number of chronic neurological disorders such as Alzheimer’s, Huntington’s and Multiple Sclerosis.
Final in-vitro results expected in coming weeks
Neurotech is positioned well to finesse its upcoming clinical program, selecting the optimum dose and patient parameters.
Commenting on the highly promising results as well as pointing to the potential for applications in other areas, Leedman said, “These preliminary trial results continue to be very encouraging, in particular the unique mode of action of our strains compared to CBD alone.”
“These results indicate that the DOLCE/NTI leads may have a broader application in relation to the management and treatment of a number of neurological disorders”.
Even with significant stock price upturn over the past month, there is potential for further share price moment as final in-vitro trial results are imminent.
The transition to clinical trials from preliminary tests is a major milestone, providing share price re-ratings as well as being the source of positive news flow throughout the trial process.
Disclaimer: This article is sponsored content from Neurotech International Limited (NTI) and does not constitute an endorsement by the publisher.


